Gene therapy has made headlines in recent years, in part because of rapid advances in biotechnology that allow doctors to perform such treatments. Broadly speaking, gene therapy is a technique used to treat or prevent disease by changing the content or expression of DNA in cells, often by replacing defective genes with functional ones.
The term “gene therapy” sometimes comes up alongside misinformation about mRNA vaccines, which include the Pfizer and Moderna COVID-19 vaccines. These vaccines contain mRNA, a genetic cousin of DNA, that prompts cells to make the coronavirus’s “spike protein.” The vaccines don’t change the cells’ DNA, and after the spike is made, the cells destroy most of the mRNA. Other COVID-19 shots include viral vector vaccines made by AstraZeneca and Johnson & Johnson, which deliver DNA to cells to prompt them to make spike proteins. Cells that make spike proteins, using instructions from either mRNA or viral vector vaccines, are targeted by the immune system so they don’t stick around long-term. This is different from gene therapy, which aims to change cell function in the long term.
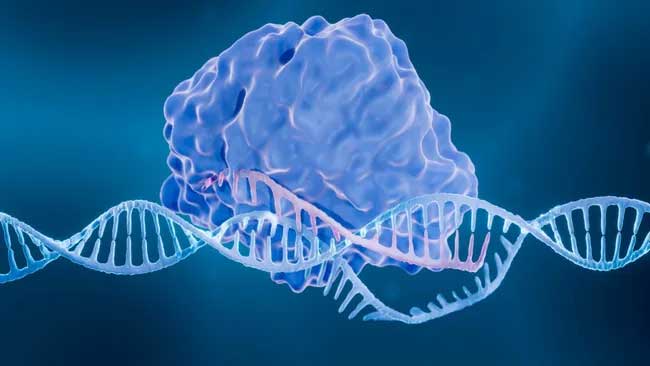
DNA is the molecule that stores genetic information, and genes are the pieces of genetic information that cells use to make a specific product, such as a protein. DNA is found inside the cell’s nucleus, where it is packaged into chromosomes, and inside the mitochondria, the “powerhouse” organelles located outside the nucleus.
While there are mitochondrial diseases that may one day be cured with gene therapy, the term “gene therapy” currently refers to treatments that target nuclear genes—genes on the 23 pairs of chromosomes inside the nucleus.
Illustration of DNA inside chromosomes, which are then located inside the cell nucleus. BSIP/UIG
Classically, gene therapy refers to the process of either “switching off” a dysfunctional gene or adding a copy of a working gene to the nucleus in order to improve cell function. Currently, gene therapy is aimed at diseases that arise from a problem with one gene, or at most a few genes, rather than those that affect many genes.
But the field of gene therapy is now expanding to include strategies that don’t all fall into the classic categories of knocking out bad genes or adding good genes. For example, researchers at Sangamo Therapeutics are developing genetic treatments for Parkinson’s, Alzheimer’s, and Huntington’s diseases that work by boosting or suppressing the activity of specific genes.
Although treatments can add genes to cells in the body, turn off genes, or change the function of genes in some way, each gene therapy targets cells in specific tissues in the body. So when scientists and doctors talk about what gene therapy does to DNA, they are not talking about all of the DNA in the body, but only a portion of it.
Gene therapy can be performed both ex vivo and in vivo.
Ex vivo gene therapy means that cells are removed from the body, treated, and then returned to the body. It is an approach used to treat genetic disorders of blood cells, as bone marrow can be taken from a patient, stem cells from that bone marrow can be treated with gene therapy – for example, to replace a gene that is missing or not working correctly – and the transformed cells can be injected back into the patient.
In vivo gene therapy means that the gene therapy itself is injected or infused into a person. This may involve an injection directly into the anatomical site where the gene therapy is needed (a common example is the retina of the eye), or it may involve an injection or infusion of the genetic cargo to be delivered to the tissues of the body where it is needed.
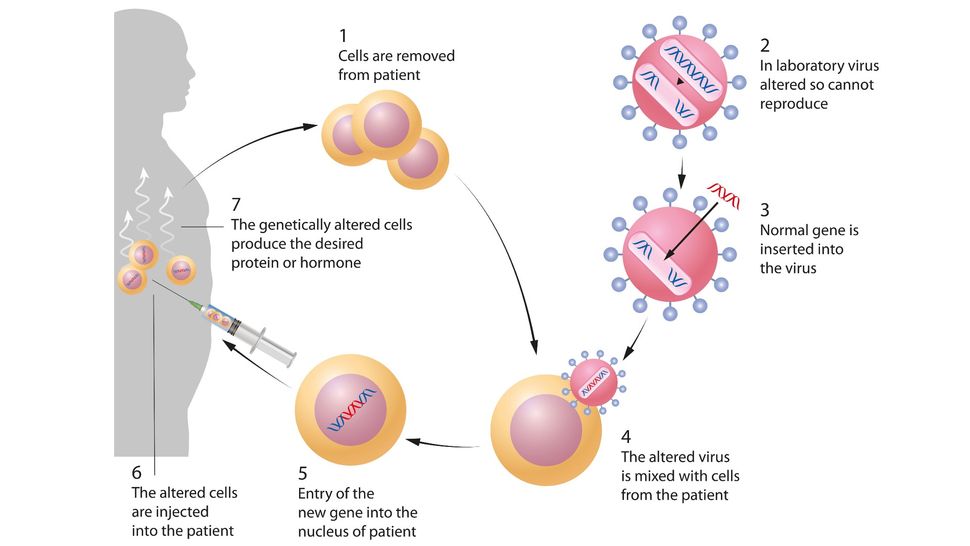
Gene therapy can involve introducing genes into human cells and tissues to treat disease. This diagram shows an example of ex vivo gene therapy. Aldona Griskeviciene
In both ex vivo and in vivo gene therapy, the genetic payload is packaged into a container called a vector before being delivered into cells or the body. One such vector is the adeno-associated virus (AAV). This is a group of viruses that exist in nature but whose normal genes have been removed and replaced with genetic payload, making them gene therapy vectors.
AAV has been used to deliver gene therapy for many years because it has a good safety record. It is much less likely to trigger a dangerous immune response than other viruses that were used as vectors decades ago when gene therapy was in its infancy. Additionally, packaging genetic payloads in AAV carriers allows injected or infused gene therapy to travel to specific tissues in the body where it is needed. This is because there are many types of AAV, and certain types are attracted to specific tissues or organs. So if the genetic payload needs to reach liver cells, for example, it can be packaged into a type of AAV that likes to go to the liver.
In the early days of gene therapy, which began in 1989, researchers used retroviruses as vectors. These viruses delivered genetic cargo directly into a patient’s nuclear chromosomes. However, there were concerns that this integration of new DNA into chromosomes could cause changes that could lead to cancer, so this strategy was initially abandoned. (More recently, scientists have successfully used retroviruses in experimental gene therapy without causing cancer; for example, retrovirus-based therapy has been used to treat infants with “bubble boy disease.”)
Moving away from retroviruses, researchers turned to adenoviruses, which offered the advantage of delivering genetic payloads in the form of episomes—a piece of DNA that functions as a gene within the nucleus but remains a separate entity from the chromosomes. The risk of cancer was extremely low with this innovation, but adenovirus vectors turned out to stimulate the immune system in very powerful ways. In 1999, an immune reaction to adenovirus-based gene therapy led to the death of 18-year-old Jesse Gelsinger, who had volunteered for a clinical trial.

Gene therapy researchers have abandoned retroviruses and switched to adenoviruses. Seksan Mongkhonkhamsao
Gelsinger’s death rocked the gene therapy community, setting the field back for several years, but the modern gene therapies that have emerged over the years based on AAV are not dangerous. However, they tend to be expensive and have variable success rates, so they are typically used as a last resort for a growing number of genetic diseases.
Gene therapy can treat some blood disorders, such as hemophilia A, hemophilia B, sickle cell disease, and, starting in 2022, beta thalassemia. What these disorders have in common is that the problem comes down to just one gene. This has made beta thalassemia and sickle cell disease easily accessible to ex vivo gene therapy, which involves removing and modifying bone marrow stem cells, while hemophilia A and hemophilia B are treated with in vivo gene therapy, which targets liver cells. However, there are other treatments for these blood disorders, so gene therapy is more of a last resort.
Numerous enzyme deficiency disorders also come down to one bad gene that needs to be replaced. Cerebral adrenoleukodystrophy, which causes fatty acids to build up in the brain, is one such disorder that can be treated with gene therapy, according to Boston Children’s Hospital. CAR T-cell therapy, which is approved for some cancers, involves removing and modifying a patient’s immune cells and is known as “cellular gene therapy.”
Gene therapy has also proven useful in treating inherited retinal diseases where other treatments have failed. Another group of gene therapy targets are diseases of the nervous system.
For example, gene therapy is being developed to treat a pair of genetic disorders called Tay-Sachs disease and Sandhoff disease. Both conditions occur when organelles called lysosomes become filled with fat-like molecules called gangliosides. The consequences of these disorders include developmental delays, loss of previously acquired skills, stiffness, blindness, weakness, and lack of coordination, with possible paralysis. Children born with Tay-Sachs disease and Sandhoff disease usually do not survive beyond the age of 2 to 5 years.
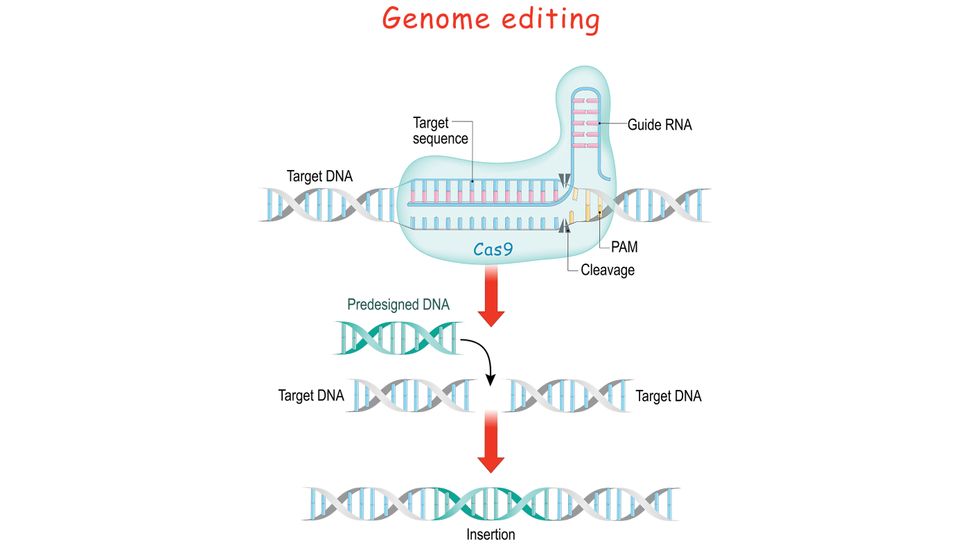
CRISPR gene editing is a powerful method of modifying DNA that could one day be used in gene therapy. Here’s a simplified description of how CRISPR gene editing works. ttsz
“There was no rational prenatal or neonatal testing for Tay-Sachs and Sandhoff syndromes because there was no treatment available,” said Dr. Jagdeep Walia, a clinical geneticist and head of medical genetics at the Department of Pediatrics and Kingston Health Sciences Centre and Queen’s University in Ontario, Canada. Walia is developing a gene therapy that aims to replace the Hex A gene, an enzyme that is deficient in these children. So far, the treatment has shown good efficacy and safety in animal models, but it still needs to be tested in humans.
The future looks bright when it comes to gene therapy in general, thanks to new technological developments, including CRISPR gene editing. This is an extremely powerful method for cutting out parts of DNA molecules and even inserting new parts. CRISPR is not the first method that scientists have used to edit DNA, but it is much more versatile than other methods. It is not quite ready for in vivo chromosome manipulation yet, but it is developing exponentially.
Perhaps even closer to the horizon is the prospect of delivering more genetic information into cells. One of the big drawbacks of the AAV vector is that each viral particle can only carry a small amount of DNA, but recent research has shown that another type of virus called cytomegalovirus can be adapted to carry gene therapy with much more useful information than AAV. Not only could this someday extend gene therapy to more diseases that require more genes than AAV can carry, but it could also allow more than one gene to be delivered in a single therapy.