The MHRA has approved Casgevy to treat sickle cell disease (SCD) and transfusion-dependent beta thalassaemia. These are lifelong genetic disorders caused by mutations in the genes that code for haemoglobin, the protein red blood cells need to transport oxygen around the body. CRISPR could treat a common form of inherited blindness. The 2020 Nobel Prize in Chemistry has gone to two women who developed a gene-editing tool called CRISPR-Cas9, which cuts DNA like molecular scissors.
Scientists introduced the world to CRISPR as a gene-editing tool in the summer of 2012, when landmark papers from two independent groups demonstrated how the system could be used to cut DNA. Now we’re seeing CRISPR used in groundbreaking medical treatments.
Before these seminal papers, other researchers had begun to unravel how CRISPR works inside microbes. Although CRISPR is best known as a gene-editing tool, it was first discovered in bacteria, and scientists realized that it acts as a kind of immune system—a defense against viruses. In this immune system, the bacterium has a memory bank full of the virus’s genetic material. The bacterium will store this material after a viral attack to protect itself from future invasions.
This memory bank is paired with tiny molecular scissors called Cas proteins that cut DNA, and a molecule that guides the scissors to their target. In bacteria, that target is a viral invader. Shikshnis and his colleagues showed that scientists can use these scissors to their advantage, targeting any DNA they want to edit. They specifically demonstrated this using the Cas9 protein.
Along with Jennifer Doudna and Emmanuelle Charpentier, the authors of another groundbreaking CRISPR paper published in 2012, Sikshnis was awarded the 2018 Kavli Prize in Nanoscience for inventing CRISPR-Cas9, a “precision nanotool for DNA editing.” He and his team are now exploring the diversity of CRISPR systems found in nature to see which ones might be useful for engineering genomes.
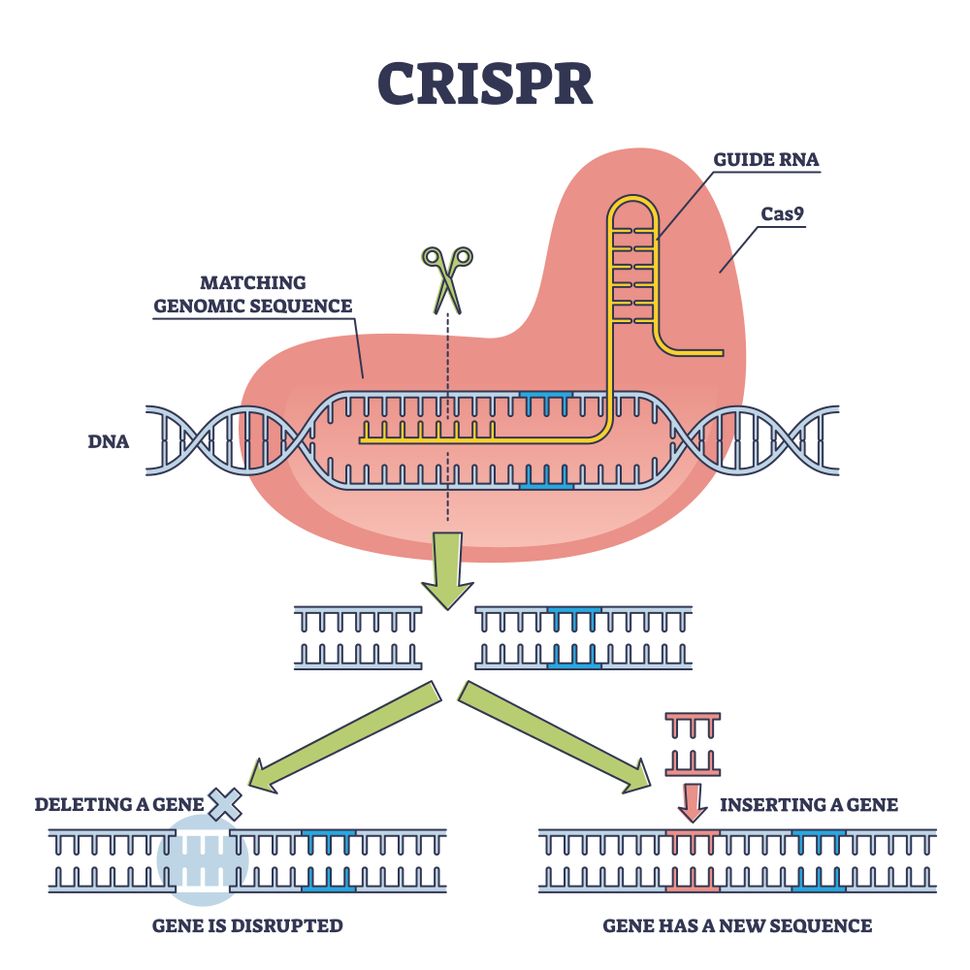
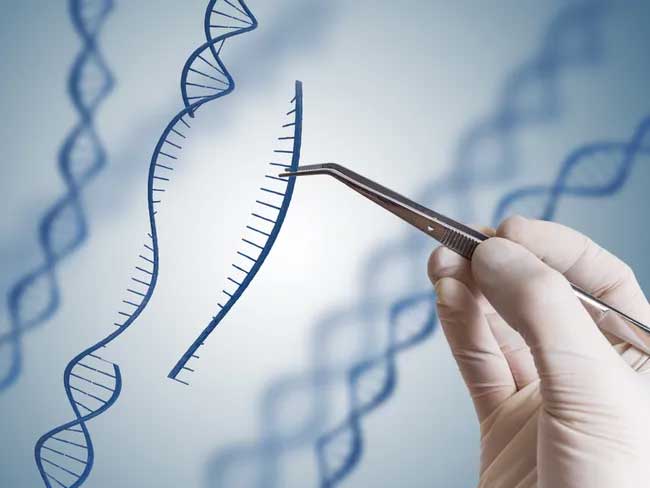
The CRISPR-Cas9 system uses Cas9 as a molecular scissors and an RNA molecule as a guide for those scissors.
A 2012 paper showed that it is possible to reprogram the Cas9 protein and target it to any sequence in the genome. This is really a kind of truly universal system that can be used for genome editing in a variety of model organisms.
There are some limitations to this treatment because in this case, the treatment is done ex vivo. This means that the cells that need to be treated are removed from the patient, and then the Cas9 tool is used to correct the mutation — or, essentially, trigger the production of fetal hemoglobin. And then these engineered cells, they have to be delivered back into the patient. And of course, this is kind of a complicated and time-consuming procedure.
Of course, it would be great if CRISPR treatment could be done directly in the human body – we call it in vivo. But in reality, to do that, we need to overcome several challenges: first, we need to deliver this CRISPR tool to specific tissues or organs in the human body. And of course, there are many ways to deliver CRISPR tools, but after COVID, mRNA vaccines have been approved as a therapeutic method for the treatment and prevention of COVID. And now, mRNA combined with lipid nanoparticles has become one of the key methods that can deliver Cas9 to various cells and tissues in the human body.
Other delivery systems are also being explored, including virus-like particles and adeno-associated viruses. So AAVs are also being used as delivery vehicles, and they are approved as safe delivery vehicles for humans – but for example, in the case of AAVs, there is a limitation on the packaging load, and smaller gene editing tools need to be found that can be packaged into a single AAV particle.

Virginijus Šikšnys studies the diversity of CRISPR systems found in nature. Vilnius University
First, researchers look at microbial DNA sequences, which are present in huge databases where new CRISPR systems can be found. They then try to express them in different bacteria, isolate them, characterize them, and then transfer them into human cells to see if they can be used as new genome modification tools.
It was clear from the start that genetic diseases caused by a single mutation, such as sickle cell disease, would be the first target. This seemed easy to achieve because it only required correcting one mutation in the genome. Part of the credit for this Cas9-based sickle cell treatment should be given to the people who have been studying sickle cell disease for decades. They provided insights into the disease mechanisms that were implicated in the treatment.
CRISPR-Cas9 is a kind of universal or multifunctional technology because it can be used to create any living organism. You’re just trying to create DNA, and DNA is the blueprint for every living organism. So instead of editing genes in human cells, you can also think about editing a population of bacteria, say, the ones that are in the human gut. And you can create these populations of bacteria.
CRISPR technologies can also be used as antiviral agents. The problem with antibiotics is pretty clear at the moment – we are probably losing our battle against bacteria using antibiotics. New antibiotics are always needed and they are really hard to find, complicated and expensive. So alternative technologies are being developed, such as viral therapy or antibacterial CRISPR systems.
CRISPR was used a few years ago in China to create human embryos – so that’s a line that scientists actually agree not to cross because it could be really dangerous.
Exa-cel, also known by the brand name Casgevy, received its first regulatory approval on November 16, 2023, from the UK Medicines and Healthcare products Regulatory Agency (MHRA) for the treatment of two debilitating blood disorders: sickle cell disease and transfusion-dependent beta thalassemia. The US Food and Drug Administration (FDA) later approved the therapy as a treatment for both conditions.
The world’s first treatment using CRISPR gene editing technology has been approved. Drug regulators have approved a CRISPR therapy called Casgevy for the treatment of inherited blood disorders.
The regulators’ historic decision to approve Casgevy could signal a new era of gene therapy. But questions remain about the treatment’s availability and long-term safety.
The MHRA has approved Casgevy for the treatment of sickle cell disease (SCD) and transfusion-dependent beta thalassaemia. These are lifelong genetic disorders caused by mutations in the genes that code for haemoglobin, the protein red blood cells need to transport oxygen around the body.
It is estimated that more than 100,000 people in the United States have SCD, but the rate is higher for some populations than others. For example, 1 in 365 black babies are born with SCD. The disease changes the shape of a person’s red blood cells so that they become C-shaped instead of round. The sickle cells quickly die and also stick together, blocking blood vessels. As a result, patients develop anemia and often have episodes of severe pain called pain crises.
Beta thalassemia affects about 1 in 100,000 people worldwide, and it disproportionately affects people of Mediterranean, Asian, African, and Middle Eastern descent. Patients with beta thalassemia do not produce enough hemoglobin, which can lead to severe anemia, while sickle cell anemia occurs due to a lack of healthy red blood cells. “Transfusion-dependent” means that the disease is so severe that patients need regular red blood cell transfusions throughout their lives.
Casgevy is based on a revolutionary gene-editing technology called CRISPR, which was first developed in 2012. The CRISPR system cuts genes from DNA using an enzyme called Cas9. These “molecular scissors” are guided to the target DNA by an RNA molecule. The technology was adapted from a natural defense mechanism that bacteria and other simple organisms called archaea use against viruses.
Casgevy targets the BCL11A gene. The gene codes for a protein that normally regulates the switch from the fetal version of hemoglobin to the adult version shortly after birth. However, in patients with SCD and beta thalassemia, the adult hemoglobin is defective.
The goal of Casgevy is to disable BCL11A and thus allow the body to continue producing fetal hemoglobin since the adult version does not work. To do this, blood-forming stem cells are taken from the patient’s bone marrow and the BCL11A gene is edited using Casgevy in the lab. The modified cells, with functioning hemoglobin, are then infused back into the patient. Before the infusion, the patient must take a chemotherapy drug called busulfan to eliminate the unedited cells still in the bone marrow, STAT News reports.
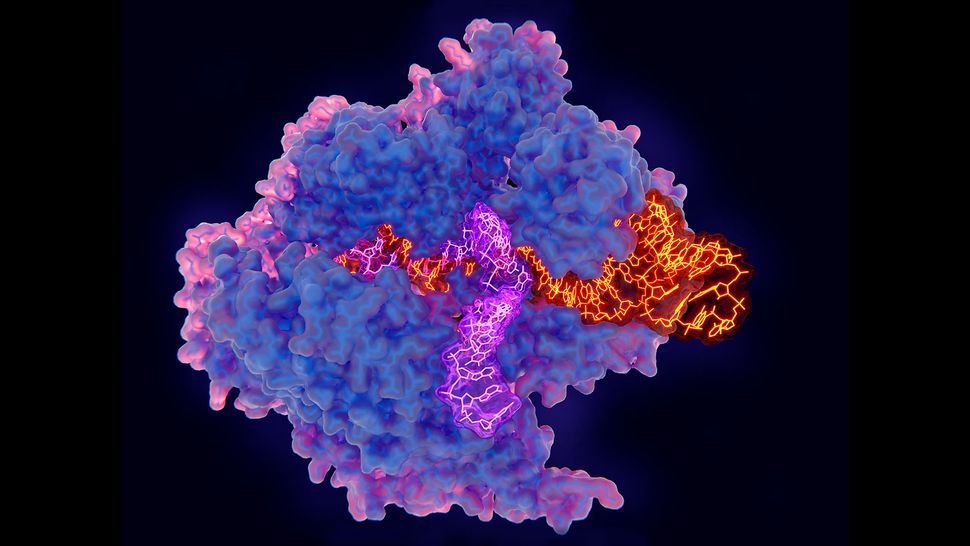
This illustration of CRISPR in action shows Cas9 (blue and pink) attached to DNA (purple) next to a guide RNA molecule (orange). JUAN GAERTNER/SCIENCE PHOTO LIBRARY
This process of adaptation to the new, edited cells is lengthy. “Patients may need to spend at least a month in hospital while the treated cells settle in the bone marrow and begin to produce red blood cells with a stable form of haemoglobin,” the MHRA said in a statement.
In two late-stage clinical trials, Casgevy restored hemoglobin production in most patients with SCD and beta thalassemia and improved their symptoms. Twenty-eight of 29 patients with SCD did not experience any major pain crises for at least a year after treatment with Casgevy. Similarly, 39 of 42 patients with beta thalassemia did not require red blood cell transfusions during the same period after treatment. The remaining three patients required transfusions more than 70% less often.
No serious safety concerns were noted in either of Casgevy’s two late-stage clinical trials, although some temporary side effects such as fever and fatigue were reported. Both of these trials are ongoing, and the long-term safety of Casgevy continues to be monitored by regulators such as the MHRA and the FDA, as well as the therapy’s makers Vertex Pharmaceuticals and CRISPR Therapeutics.
However, there are still some concerns about the safety of CRISPR-based therapies in general. Namely, there are concerns about “off-target” effects, which occur when Cas9 acts on other parts of the genome that it was not supposed to change and causes unwanted side effects.
“It’s well known that CRISPR can produce spurious genetic modifications with unknown consequences for the treated cells,” David Rueda, head of molecular and cellular biophysics at Imperial College London, told the UK Science Media Centre. “We need to see whole-genome sequencing data from these cells before we can draw any conclusions,” he said. This would involve examining all the DNA in Kasgevi-edited cells to see if there are any off-target effects.
In November 2023, the U.K. approved Casgevy for people 12 years and older with sickle cell disease or transfusion-dependent beta thalassemia. In December, the FDA approved the treatment for people 12 years and older with sickle cell disease, and in January 2024, the agency approved Casgevy for people with transfusion-dependent beta thalassemia in the same age category.
The treatment is currently being reviewed by the European Union’s European Medicines Agency and Saudi Arabia’s Food and Drug Administration, so other countries may soon approve Casgevy, according to Vertex.
It’s unclear when Casgevy will become available, but its reach will depend largely on its cost. Gene therapies can cost millions of dollars, and it’s likely that Casgevy will be no exception. That could put it out of reach for many people who need it.
“The problem is that these treatments will be very expensive, so the key is to make them more widely available around the world,” Kay Davies, professor of anatomy at the University of Oxford, told the UK Science Media Centre.
A Vertex spokesperson told Nature that the company has not yet set a price for Casgevy in the UK but is “working with health authorities to ensure reimbursement and access to the drug for eligible patients as soon as possible.”
Intellia Therapeutics is developing CRISPR therapies to treat inherited diseases from within the body, STAT News reports. Additionally, a modified version of CRISPR called “base editing,” which can target individual building blocks of DNA, is being tested as a way to treat diseases. For example, Verve Therapeutics is testing such an experimental treatment for heart disease. Another promising new type of therapy, called “base editing,” involves CRISPR but also “includes additional enzymes and genetic instructions to insert, delete, or rewrite short segments of DNA,” STAT News reports.
CRISPR may treat a common form of inherited blindness, early evidence suggests. In a small study, some people with inherited vision loss saw their vision improve after treatment with CRISPR.
Early clinical trials suggest that CRISPR therapy injected directly into the eye may show promise in treating the most common form of inherited vision loss in children.
This form of vision loss, called Leber congenital amaurosis (LCA), is often apparent at birth and results from dysfunction or death of light-sensing cells called photoreceptors in the retina, at the back of the eye. Such problems are caused by mutations in any one of at least 20 genes.
Some of the most common causes of LCA are mutations in the gene that codes for centrosomal protein 290 (CEP290). More than three-quarters of people with the condition carry a specific mutation that affects CEP290, which is critical for the proper functioning of photoreceptors.
There is currently no cure for LCA — but there is now evidence that the well-known gene-editing tool CRISPR can be safely used to improve the vision of some people with the condition. The results of an early-stage study were published in The New England Journal of Medicine.
The trial is also notable because it involved the first person to ever receive a CRISPR-based treatment directly in the body. By comparison, the first approved CRISPR therapy involves removing cells from the body, editing them in the lab, and then returning them to the patient.
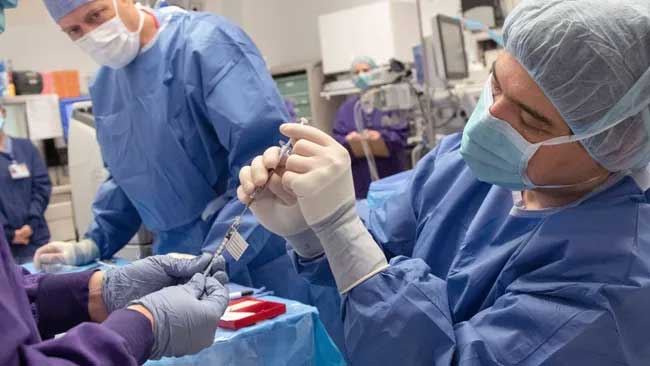
Here, surgeons at OHSU’s Casey Eye Institute perform the CRISPR gene-editing procedure in the body as part of a recent clinical trial. OHSU/Kristyna Wentz-Graff
The study involved 14 people — 12 adults and two children. All had a specific mutation in the CEP290 gene that affects most LCA patients. The participants received a single injection of a CRISPR drug called EDIT-101 into the eye with the most significant vision loss. The other eye served as a comparison.
EDIT-101 contains tiny guides that guide pairs of “molecular scissors” — called Cas9 enzymes — to the mutant CEP290 gene. The scissors cut out the defective portion of the gene, thereby restoring its function.
The team used a CRISPR-based strategy because CEP290 is a large gene, making it a difficult target for traditional gene therapy. Some gene therapies use modified viruses to deliver functional genes into cells to replace defective genes, but the CEP290 gene is too large to fit into such a delivery system.
Following this treatment, all participants underwent vision testing, which was performed every three months for a year, followed by less frequent monitoring for two years. By the end of the trial period, 11 of the 14 volunteers showed measurable improvements on at least one vision test, while six experienced improvements on two or more tests. One trial participant reported being able to find his phone if he lost it, and was able to see the small lights on his coffee machine, something he had not been able to do before the treatment.
Those who showed no measurable improvement tended to be at a more advanced stage of the disease, in which their cells showed high levels of dysfunction at baseline, the study participants noted. None of the participants experienced adverse side effects from the treatment.
While EDIT-101 can treat cells that are present in the retina, it cannot reverse the loss of cells that have already died. This means that participants may experience some improvement in their vision, but it remains reduced.
The next step will be to test the therapy in more patients. The team is particularly hoping to test the drug in younger patients, who may hopefully have even better results.
The 2020 Nobel Prize in Chemistry has gone to two women who developed a gene-editing tool called CRISPR-Cas9 that cuts DNA like molecular scissors.
The technology “has not only revolutionized basic science, but has also led to innovative crops and will lead to groundbreaking new treatments,” Nobel Chemistry Committee Chairman Claes Gustafsson said in a statement. By being able to expertly cut specific DNA sequences from the genome, scientists can pinpoint gene functions; these discoveries not only add to our basic understanding of how these genes work, but could also have practical applications, such as developing drought- and pest-resistant crops and developing treatments for cancer and genetic diseases. The genetic cut-and-paste system is also being used in new COVID-19 diagnostic tests.
The Nobel Prize “for the development of a method for genome editing” was awarded to Emmanuelle Charpentier, director of the Max Planck Society’s Pathogens Unit, and Jennifer Doudna, professor of biochemistry, biophysics, and structural biology at the University of California, Berkeley. According to Science Magazine, this is the first Nobel Prize in science to be awarded to an all-female team.
The development of CRISPR-Cas9 began serendipitously when Charpentier was studying the bacterium Streptococcus pyogenes, which causes a range of illnesses from tonsillitis to sepsis, according to a statement from the Nobel committee. The bacteria contain a molecule called tracrRNA, Charpentier discovered, that protects S. pyogenes from infection by viruses, according to a 2011 report in the journal Nature.
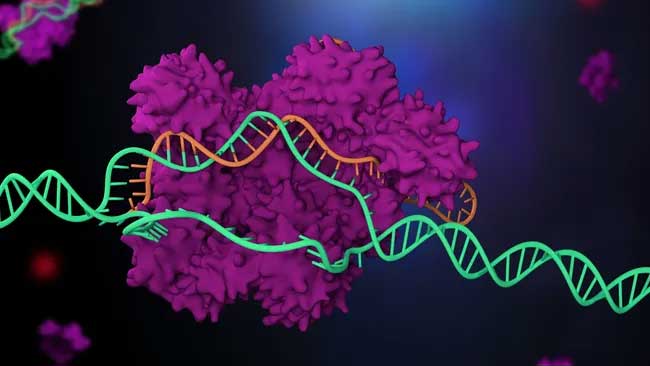
It turns out that tracrRNA is just one component of a larger defense mechanism known as the CRISPR/Cas system, which bacteria use to cut and cleave the DNA of viruses that try to infect them. After a viral attack, bacteria incorporate some of the viral DNA into their own genome; these battle trophies line up repeatedly throughout the genome and are known as “clusters of regularly interspaced short palindromic repeats,” or CRISPR for short. These archived genes are thought to help bacteria recognize viruses and fend off future attacks.
But to first cut the viral DNA, the bacteria use “CRISPR-associated” proteins called Cas proteins, under the guidance of tracrRNA and other molecules.
After discovering tracrRNA, Charpentier began collaborating with Doudna, and the two recreated the bacteria’s genetic scissors in a test tube. In their seminal work, published in 2012 in the journal Science, they simplified the system into a usable gene-editing tool capable of targeting and cutting specific DNA sequences from the genome. Since then, the tool has been further refined and used for a wide range of applications, including the recent development of diagnostic tests for COVID-19, the BBC reports.
“This discovery, originally derived from a natural defense mechanism of bacteria against viruses, will have countless applications in treating and curing genetic diseases and fighting cancer, and will also impact agriculture and other areas,” Louis Echegoyen, president of the American Chemical Society, said in a statement. “The future of this technology is truly bright and promising.”
Some scientists expected Broad Institute biochemist Feng Zhang to share the Nobel Prize with Charpentier and Doudna because he demonstrated shortly after their discovery that CRISPR also works in mammalian cells, Science Magazine reported. Based on Zhang’s work, the Broad Institute received the first patent for using CRISPR gene-editing technology in eukaryotes — complex cells with nuclei to store DNA — but Charpentier and Doudna’s institutes continue to fight for their own patents, according to The Scientist.