Layers of methane are locked under permafrost in Arctic regions, but if they were released it could cause problems for the rest of the world. Researchers also discovered intense methane emissions deep in the Baltic Sea. Methane can be a real problem for the environment. The greenhouse gas, made up of carbon and hydrogen and about 80 percent more potent than carbon dioxide, occurs naturally in sedimentary layers of the seafloor and is responsible for about 30 percent of global temperature rise.
Methane in the Arctic
Researchers believe that subglacial methane in the Arctic could amount to several million cubic feet, and its release could have serious environmental consequences. Beneath the expanse of Arctic permafrost lurks a climate-changing sea of methane. It is trapped under layers of ice and slowly leaks out when cracks appear. Any sudden change in the permafrost could release what researchers estimate to be several million cubic feet of environmentally damaging gas.
Any large-scale gas leak could trigger a potentially destructive warming loop: methane would cause further melting of the permafrost, which would then lead to additional gas emissions. By studying the permafrost (any ice that remains stable for two or more years is considered permafrost) beneath the Svalbard islands, researchers discovered that this layer is not in a permanent state. They also discovered that huge amounts of methane, normally trapped below, were able to migrate even while remaining trapped.

Using historical downhole monitor data, the team observed a continuing trend of gas accumulating at the base of the permafrost. But this was not a uniform finding. Sometimes, when the team thought locations were obvious sites for gas, they found no gas, assuming it had already migrated. Other places were so full of gas that the team drilling the well could hear explosive levels of methane bubbling.
The study was conducted in Norway, but because the region’s geological and glacial history is similar to the rest of the Arctic region, the team believes that migrating methane deposits are likely present elsewhere. The permafrost that contains methane is not homogeneous and continuous, and neither is the safety of these methane deposits.
Ocean currents can thin permafrost and create uneven densities. The highlands are drier and more permeable, while the lowlands have permafrost that is saturated with ice. And researchers say that even where continuous permafrost exists, geographic features may allow gas produced by the rocks below to escape. Because permafrost conditions are constantly changing, it may only be a matter of time before methane emissions increase.
Methane in the Baltic Sea
Methane is leaking from the Baltic Sea. The leak, located in the Londsort Trench, is causing gas to seep across more than seven square miles of seabed about 18 miles off the Swedish coast, leaking gas into the water. What’s unusual is not only the depth of the find—about 1,300 feet—but also the fact that gas bubbles almost all the way to the surface.
Marcelo Coetzer, professor of environmental sciences at Linnaeus University, believes that deep ocean currents are causing the accumulation of methane-rich sediments in this particular area. In any case, this does not explain how powerful the methane bubbles are, which remain high in the water column while they float. The leak in the Londsort Deep is powerful, with bubbles rising more than 1,200 feet from the bottom as monitored by sonar.

Although incredibly deep methane emissions can freeze the bubbles, allowing them to remain intact longer, freezing does not occur in the Baltic Sea. Why the bubbles are so powerful and stable is still unknown. The team believes the anoxic environment of the Baltic Sea may be the cause. “If there is no oxygen, the levels of dissolved methane in the ocean can be relatively high, which in turn causes the bubbles to not lose methane as quickly.” Thus, the bubbles in this environment remain more intact, meaning transport of methane to the sea surface becomes more efficient.
The team argues that if oxygen conditions in the Baltic Sea deteriorate further, this will likely lead to increased transport of methane from deeper parts of the sea. From there, this methane can enter the atmosphere.
Absorption of carbon dioxide by the ocean and artificial removal of carbon from the ocean
The ocean is a huge carbon sink. Research shows that it has absorbed more than a quarter of all the carbon dioxide that people put into the atmosphere. However, scientists have found that even the ocean is reaching its limit and could become only half as efficient at sequestering carbon by 2300 due to extreme warming and acidification. But a growing number of startups, including Captura and Running Tide, are promising to help reduce the ocean’s carbon load by using technology to suck greenhouse gases directly from seawater.
In one of its biggest projects, Equatic and Singapore’s national water agency PUB have announced they will build the world’s largest ocean carbon removal facility. Last year, the company, owned and operated by UCLA scientists, developed two pilot projects in Los Angeles and Singapore that successfully extracted about 100 kilograms (0.1 ton) of carbon dioxide every day. Now their goal is to build a $20 million plant that will remove 3,650 metric tons (4,000 tons) of gas from the ocean annually, according to a UCLA press release.

How it works? In the Equatic Process, operators begin by passing an electrical current through seawater pumped into the site. This charge splits water (H2O) into hydrogen and oxygen, then air passes through the water, which traps and stores the CO2 in calcium- and magnesium-based solids, “similar to how seashells form naturally,” Time magazine reported. Removing carbon from water allows the ocean to absorb more carbon from the air, the company says. In addition to storing CO2, the company plans to use the hydrogen byproduct as an ingredient in clean fuels, according to the statement.
In September, more than 200 experts signed a letter outlining their concerns: “While ocean-based approaches to carbon removal have enormous potential, there are also risks,” they wrote. “The public does not yet have sufficient information about the effectiveness or impact of any particular approach and therefore cannot make informed decisions about their use on a large scale.”
In May 2023, a UN commission called the carbon capture industry “unproven” with “unknown” risks and questioned its economic viability due to high operating costs. But the US federal government is certainly betting big on research that will help improve the ocean’s ability to remove carbon from the atmosphere.
In October 2023, the Department of Energy (DOE) allocated $36 million to “develop methods for removing carbon dioxide from the marine environment.” The National Science Foundation has taken the first steps toward launching a federal research program to study various methods, such as using “minerals and electricity to change the alkalinity of the oceans to allow seawater to absorb more CO2 from the air,” E&E News reports.
Intentional stratospheric dehydration to remove water vapor from the atmosphere
While anthropogenic carbon dioxide emissions are by far the most important driver of climate change, water vapor is actually the most abundant greenhouse gas and is responsible for about half of Earth’s natural greenhouse effect – the one that makes our planet habitable.
Now, as scientists explore ways to reverse the effects of climate change by removing excess heat-trapping carbon dioxide from the atmosphere and reflecting sunlight back into space, one group of researchers wondered: Could removing some water vapor from the atmosphere also help mitigate the effects of climate change? This is exactly the idea explored in a new paper published in the journal Science Advances, which the authors call “intentional stratospheric dehydration” or PSD.
Researchers at NOAA’s Chemical Sciences Laboratory have developed the concept of PSO, which involves the dispersal of fine particles—small ice crystals—in high layers of the atmosphere that are both very cold and saturated with water vapor. Pure water vapor does not form ice crystals easily.
It is necessary to have certain growth points or condensation nuclei, for example, dust particles, around which excess moisture would condense. If such nuclei could be introduced into supersaturated air masses that are directed into the stratosphere, then some of the water vapor in this air will condense into ice and fall out as precipitation, thereby removing excess water vapor and dehydrating (at least partially) the stratosphere.

The basic physical principles behind this concept are simple in theory, but does such a region of the atmosphere exist? Indeed, there is, and it has a name: the Western Pacific Cold Spot, an area of the atmosphere roughly the size of Australia. The main route of water vapor penetration into the stratosphere is upward transport through the tropopause – the boundary separating the troposphere from the stratosphere – in the tropics.
The tropopause over the tropical western Pacific is known to be a critical factor in determining the amount of water vapor that is transported into the stratosphere. At this point it is cold enough to naturally dry out moist air through the formation of ice crystals. The problem is that pure water vapor does not form ice crystals so easily, and there are quite a few available condensation nuclei in the air. Without their presence, the relative humidity of the air relative to the ice must be about 200% for ice crystals to spontaneously form.
In their study, the scientists used a computer model to simulate conditions in the stratosphere over the Western Pacific Ocean, based on observations of the temperature and movements of tropical air near the stratosphere. This showed that the PSO concept was effective in theory.
They also analyzed high-resolution measurements of water vapor and temperature collected by NASA’s ATTREX (Airborne Tropical TRopopause EXperiment) mission in 2014. The Guam-based ATTREX mission used NASA’s Global Hawk unmanned aircraft to study the tropical tropopause over the tropical Pacific Ocean. ATTREX measurements showed that of the 550 areas of moisture-laden air encountered by Global Hawk, only 10% were in areas where water vapor could be removed by PSO.
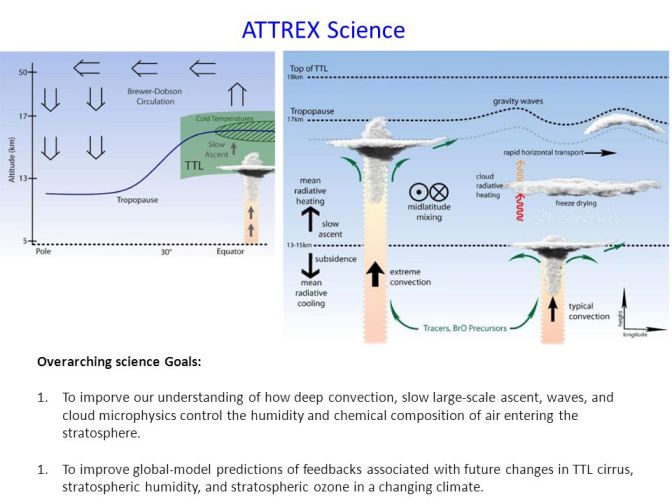
Based on this result, the authors found that the climatic effect of ice PSO is possible only in the most moisture-saturated air. The resulting magnitude of stratospheric water depletion would equate to a reduction in radiative forcing of about 1/70th that of anthropogenic CO2 emissions since 1750. This is a very small effect, scientists say, and that the PSO effect alone would not be able to counteract much of the warming caused by CO2.
However, PSO may be valuable as one element in a broader portfolio of climate change intervention and mitigation strategies because all of the techniques being studied (e.g., stratospheric aerosol injection and marine cloud brightening) have different positive and negative outcomes and different time frames. efficiency framework. All of these factors go into deciding whether a method is worthy of further study.
If decisions about climate intervention are needed, it will be critical for scientists to adequately study both all the methods by which humanity might change the climate, and the broader consequences of those methods.